Europe TIC Market for Pharmaceuticals and Biotech Industry Size & Share - Growth Forecast Report (2026-2035)
Industry Insight by Service Type (Testing Services, Inspection Services, Certification Services), by Sourcing Type (Outsourced Services, In-house Services), by Industry Vertical (Biopharmaceutical & Biologics Analysis, Environmental Monitoring, Extractables & Leachables Studies, Sterile Compounding, Trace Metal Services, Material Sciences Testing), by End User (Pharmaceutical Manufacturers, Biotech Companies, Contract Research Organizations, Academic & Research Institutes)
| Status : Published | Published On : Mar, 2026 | Report Code : VRSME9200 | Industry : Semiconductor & Electronics | Available Format :
|
Page : 146 |
Europe TIC Market for Pharmaceuticals and Biotech Industry Overview
The Europe TIC Market for Pharmaceuticals and Biotech Industry which was valued at approximately USD 3.2 billion in 2025 and is estimated to rise further up to almost USD 3.5 billion by 2026, is projected to reach around USD 7.9 billion in 2035, expanding at a CAGR of about 8.1% during the forecast period from 2026 to 2035.
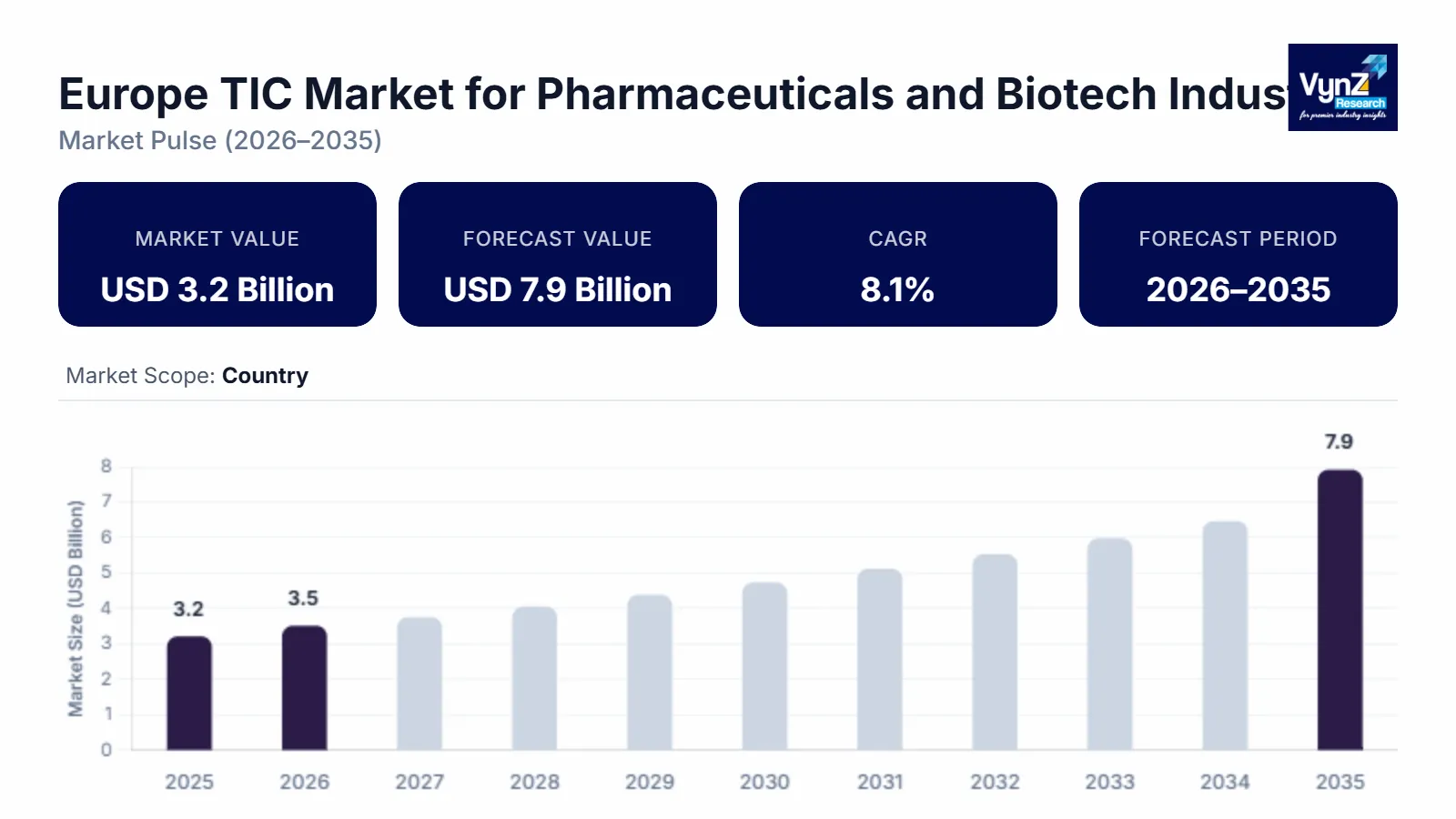
The market experiences expansion because pharmaceutical manufacturing facilities need to meet growing regulatory compliance standards while manufacturers handle more complicated biologics and advanced therapeutic products and drug development processes need improved quality assurance standards, which outsourcing testing and certification services has become more common. The market expansion throughout major regions, which include Germany, the U.K. and France, receives additional support from the increasing need for approved pharmaceutical products according to established standards and the continuous funding for regulatory harmonization and laboratory infrastructure development.
The market receives additional support from institutions that establish strict product safety and efficacy requirements through their public health oversight systems. Regulatory authorities implement inspection and certification procedures based on World Health Organization and European public health agency guidelines to enhance their oversight of pharmaceutical and biotech products. The demand for third-party testing services experiences growth because manufacturers increase their focus on good manufacturing practices while implementing strict validation requirements for biologics and sterile products. The government-backed initiatives that promote pharmaceutical innovation and clinical research expansion and cross-border regulatory alignment strengthen the TIC services in regional healthcare systems, which results in sustained market growth for the sector.
Europe TIC Market for Pharmaceuticals and Biotech Industry Dynamics
Market Trends
The pharmaceutical sector in Europe is facing major changes as its regulatory system and technological advancements affect its quality control processes. The market experiences its most significant change through companies which now choose to use testing and certification services that outside organizations provide since this approach helps them achieve better operational efficiency and standardization of their compliance procedures and reduction of their expenses. The WHO together with European public health authorities require RO regulatory frameworks to adopt their stringent safety and validation protocols which directly lead to increased need for specialized analytical and inspection service solutions.
The current trend identifies as technology develops and regulatory frameworks change companies now use digital and automated testing systems. Companies now use laboratory platforms which operate through real-time data monitoring systems to drive their transition towards integrated service models which deliver their most valuable services. The European Union establishes standardized TIC solutions through its government-supported programs which promote both pharmaceutical innovation and regulatory harmonization throughout Europe.
Growth Drivers
The market growth depends mostly on rising regulatory pressure which requires pharmaceutical and biotech companies to meet their compliance obligations. The market development process speeds up because companies spend more money on drug manufacturing infrastructure and research facilities. Public health bodies dedicated to World Health Organization guidelines now develop inspection and certification standards which require third-party TIC services. The advanced therapeutics treatment field requires specialized testing services due to its rising demand and currently existing complex biologics. Pharmaceutical companies will keep needing complete TIC solutions which government programs support through clinical research and drug innovation initiatives.
Market Restraints / Challenges
The market has attractive growth opportunities yet it suffers from two major obstacles which stem from its complex regulatory system and expensive need for compliance. The adoption of multi-country standards becomes difficult for smaller businesses because they must meet strict validation standards. European public health authorities report that all biologics and sterile products need substantial financial resources to maintain regulatory compliance. The operational process of the organization encounters difficulties because it requires both highly skilled personnel and advanced laboratory equipment. Testing equipment expenses combined with the need to purchase international technology create financial challenges which restrict market expansion during times of economic instability.
Market Opportunities
Advanced biologics and specialized testing services create substantial market prospects because pharmaceutical companies innovate new products while high-precision analytical solutions remain unmet. Customizable TIC services which deliver exceptional performance to clients will benefit from rising biopharmaceutical industry demand. European research and development initiatives which receive government funding now create business opportunities in the market. Digital and automated TIC solutions create another main business opportunity since smart laboratory systems and data-driven platforms now enhance efficiency while improving service delivery. Artificial intelligence testing systems and integrated compliance systems will enhance operational accuracy which public investment will support through regulatory modernization and pharmaceutical innovation initiatives.
Europe TIC Market for Pharmaceuticals and Biotech Industry Report Coverage
|
Report Metric |
Details |
|
Historical Period |
2020 - 2024 |
|
Base Year Considered |
2025 |
|
Forecast Period |
2026 - 2035 |
|
Market Size in 2025 |
USD 3.2 Billion |
|
Revenue Forecast in 2035 |
USD 7.9 Billion |
|
Growth Rate |
8.1% |
|
Segments Covered in the Report |
By Service Type, By Sourcing Type, By Industry Vertical, By End User |
|
Report Scope |
Market Trends, Drivers, and Restraints; Revenue Estimation and Forecast; Segmentation Analysis; Companies’ Strategic Developments; Market Share Analysis of Key Players; Company Profiling |
|
Regions Covered in the Report |
Germany, The United Kingdom, France, Rest of the World |
|
Key Companies |
ALS Limited, Applus+, Bureau Veritas, DEKRA SE, Eurofins Scientific, Intertek Group plc, SGS SA, The British Standards Institution, TÜV SÜD, UL LLC |
|
Customization |
Available upon request |
Europe TIC Market for Pharmaceuticals and Biotech Industry Segmentation
By Service Type
The market reached its highest testing services revenue in 2025 when testing services generated approximately 58% of total market revenue. Their market leadership exists because their technology solutions find usage in pharmaceutical quality control, biologics validation and all regulatory compliance requirements. The European Union has intensified its safety and efficacy standard enforcement together with WHO guideline compliance, which has led to increased adoption of these standards. The increasing demand for analytical testing which occurs during drug development and manufacturing processes will drive growth for this field which will experience a compound annual growth rate of 8.3% throughout the projection period.
The sector for inspection and certification services will maintain its growth pattern while certification services will become the sector with the highest growth rate which will achieve 8.6% annual growth between 2026 and 2035. The need for standardized compliance in cross-border pharmaceutical trade, together with increasing regulatory harmonization, drives this market expansion. The two factors of increased clinical trial activity and contract manufacturing operations throughout Europe lead to heightened demand of inspection services which guarantee product quality control while meeting regulatory requirements in different geographic areas.
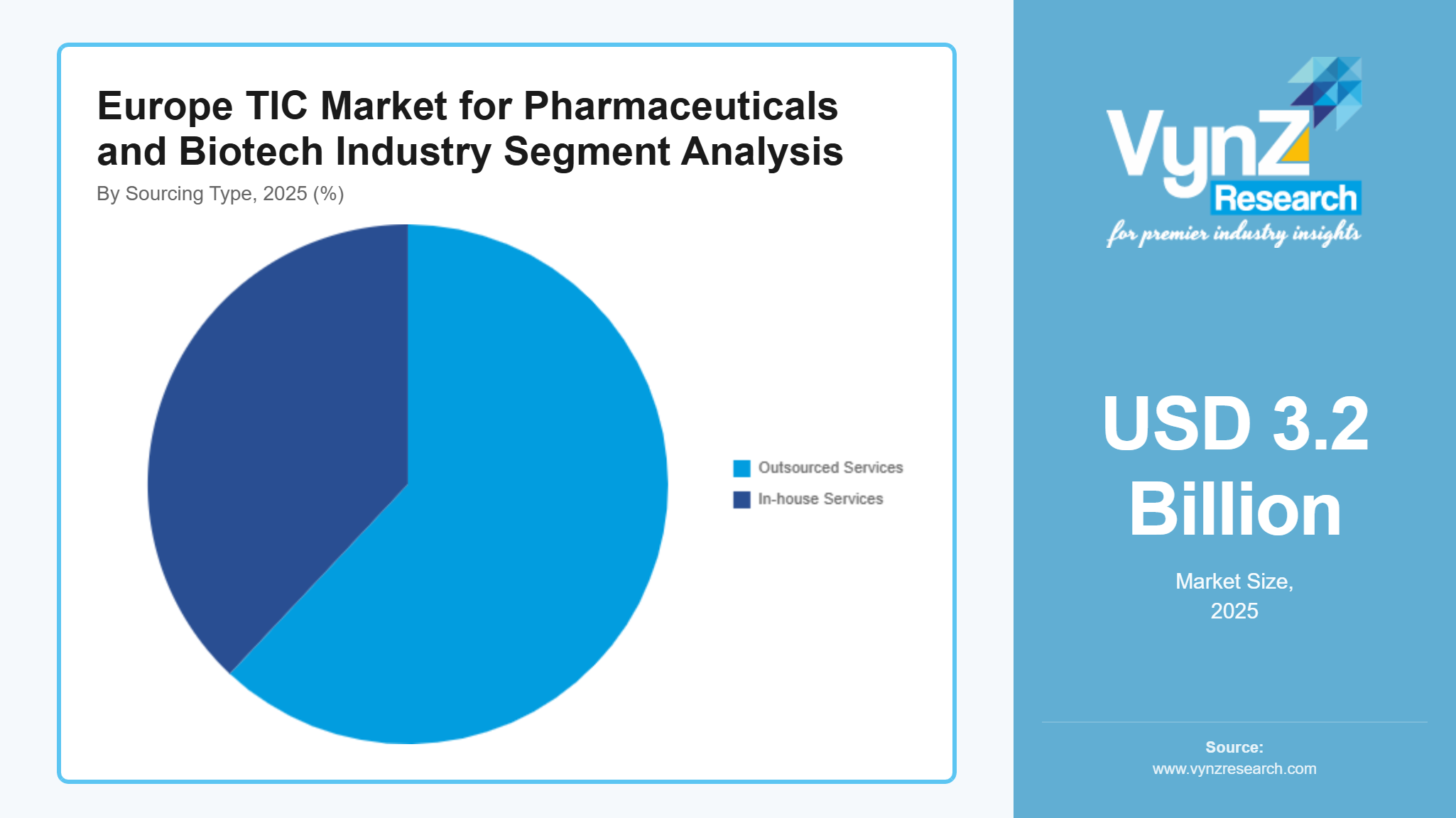
By Sourcing Type
Outsourced services held the dominant share in 2025, contributing approximately 62% of total market revenue, supported by cost efficiency, access to specialized expertise, and scalability advantages. The pharmaceutical and biotech sector now depends on third-party TIC providers for their regulatory needs and advanced testing requirements. The European Union promotes laboratory outsourcing through its government-funded research programs and regulatory framework which predicts an 8.4% compound annual growth rate for this sector during the upcoming years.
Businesses with in-house services, retained their value within large drug companies, which projected 7.2% annual growth until 2025. The segment experiences growth because internal quality control protocols require ongoing monitoring during all stages of drug development. The operational costs which continuously rise and the advanced analytical needs of business operations, particularly among small and mid-sized biotech firms, lead organizations to prefer outsourcing instead of maintaining operational expenses.
By Industry Vertical
Biopharmaceutical and biologics analysis accounted for the largest share in 2025, representing nearly 28% of total segment revenue. The industry leads the market because increasing numbers of complex biologics and biosimilars require special testing and validation for advanced therapeutics. European regulatory bodies, which operate under global health organization frameworks, require high quality and safety standards for biologic products, which leads to sustained market growth at an estimated compound annual growth rate of 8.5%
Environmental monitoring and extractable and leachable studies are expected to witness the fastest growth, with a CAGR of approximately 8.8% during the forecast period. Growth occurs because pharmaceutical manufacturers need to control contamination, produce sterile materials and ensure their materials are compatible with different manufacturing processes. The market has expanded through various applications which include sterile compounding, trace metal services and material sciences testing, which have all experienced increased regulatory oversight and quality assurance needs throughout the pharmaceutical value chain.
By End User
Pharmaceutical manufacturers accounted for the largest share of the market in 2025, contributing approximately 57% of total revenue. The company maintains market leadership through its drug production activities, required regulatory compliance and expanded scoring in drug development and production facilities. The demand for TIC services in this sector continues to grow because government-supported healthcare programs together with regulatory frameworks in Germany, the U.K. and France all support this trend.
Biotech companies and contract research organizations are expected to register the fastest growth, with a CAGR of approximately 8.7% from 2026 to 2035. The market experiences growth because multiple factors exist, which include innovation development in biologics and the ongoing clinical trial expansion and testing and certification service outsourcing. Academic and research institutes provide continuous demand through their public funding programs which support joint research efforts to develop new pharmaceutical and biotechnology innovations throughout Europe.
Regional Insights
Germany
Germany will make up 27% of the market in 2025 as its pharmaceutical manufacturing base and laboratory capabilities and European regulatory frameworks combine to create effective market conditions. The pharmaceutical and biotech companies in Berlin, Frankfurt and Munich depend on testing and inspection and certification services which these major cities provide. The existence of government healthcare programs together with their compliance to World Health Organization and European public health agency guidelines creates an environment which improves quality assurance practices. The market shows continuous expansion because companies who invest in biologics production and clinical research and pharmaceutical export activities will start using advanced TIC services.
United Kingdom
The United Kingdom held 19% of the regional market in 2025 because its established life sciences industry and its extensive clinical research network support market activities. The cities of London, Cambridge and Manchester function as main pharmaceutical innovation sites which create demand for TIC services in research and manufacturing operations. The market growth is being driven by government programs which improve drug safety and ensure regulatory compliance while funding research activities. Public health frameworks and alignment with global standards continue to encourage investments in advanced testing and certification services, which improve market outcomes.
France
France held 14% of the 2025 market because its pharmaceutical industry expands and the country develops biologics and advanced therapeutics. The cities of Paris and Lyon experience growth because their laboratory infrastructure modernization process meets the increasing need for high-precision testing services. The European standards of government healthcare systems and their regulatory frameworks create a framework which encourages pharmaceutical companies and research institutions to adopt TIC services. The country's drug development investments and quality assurance process investments have resulted in sustained market growth throughout the nation.
Rest of Europe
The Rest of Europe holds 18% of the 2025 market because pharmaceutical production activities increase and regulatory systems become better in Italy, Spain and Russia. The TIC services market experiences continuous demand because laboratories expand their facilities and more companies choose to implement quality assurance systems. The pharmaceutical industry grows in the region because government-backed initiatives which advance research and development, regulatory compliance and international standards development create a favorable environment. The smaller European countries which distribute the remaining market share of Germany and United Kingdom and France and Rest of Europe show gradual market growth which originates from their developing healthcare systems and regulatory system improvements.
Competitive Landscape / Company Insights
The market in Europe operates with moderate competition because established international and domestic companies compete through their development of new services and their expertise in regulatory matters and their business operations across different regions. Companies are investing in laboratory infrastructure and digital testing platforms and quality management systems to strengthen their market position. The existing regulatory frameworks that match World Health Organization standards and European public health authority standards enable companies to develop compliance-based services which improve their credibility while expanding their business operations throughout the region.
Mini Profiles
ALS Limited focuses on analytical testing and laboratory services, supported by strong global laboratory networks, regulatory expertise, and cost-efficient operations enabling reliable quality assurance across pharmaceutical and biotech applications.
Bureau Veritas operates in compliance and certification segments, emphasizing performance, regulatory adherence, and global inspection capabilities, supported by strong brand recognition and extensive presence across pharmaceutical and life sciences industries.
DEKRA SE leverages inspection, certification, and digital testing solutions to expand market presence, supported by strategic partnerships, technical expertise, and strong compliance frameworks across regulated pharmaceutical and biotech environments.
Eurofins Scientific focuses on advanced laboratory testing and bioanalytical services, supported by extensive global infrastructure, strong research capabilities, and specialization in pharmaceutical, biotech, and clinical testing solutions.
Intertek Group plc operates in quality assurance and certification segments, emphasizing performance, compliance, and integrated solutions, supported by strong global distribution networks and expertise in pharmaceutical regulatory standards.
Key Players
- ALS Limited
- Applus +
- Bureau Veritas
- DEKRA SE
- Eurofins Scientific
- Intertek Group plc
- SGS SA
- The British Standards Institution
- TÜV SÜD
- UL LLC
Recent Developments
In February 2026, SGS reported strong full‑year 2025 results, with rising demand for pharmaceutical and biotech testing services. The company confirmed ongoing investments in laboratory infrastructure and quality assurance capabilities for 2026.
In April 2026, Intertek highlighted continued growth in digital compliance and inspection services for regulated industries. The company is accelerating integration of digital platforms to enhance testing and assurance capabilities.
In March 2026, Bureau Veritas released its 2025 Universal Registration Document, emphasizing strategic investments in high‑value inspection and certification services. Focus remains on digital solutions and analytics to meet complex compliance requirements.
In August 2025, Eurofins expanded laboratory testing services across pharmaceuticals and biotech, strengthening its global life sciences footprint. Market reports highlighted its technical expertise and broad service offerings as key differentiators.
In July 2025, Applus+ enhanced compliance and inspection offerings for regulated sectors, supporting growth in pharmaceuticals and biotech testing. The company expanded its laboratory service capabilities internationally to meet increasing demand.
Europe TIC Market for Pharmaceuticals and Biotech Industry Coverage
Service Type Insight and Forecast 2026 - 2035
- Testing Services
- Inspection Services
- Certification Services
Sourcing Type Insight and Forecast 2026 - 2035
- Outsourced Services
- In-house Services
Industry Vertical Insight and Forecast 2026 - 2035
- Biopharmaceutical & Biologics Analysis
- Environmental Monitoring
- Extractables & Leachables Studies
- Sterile Compounding
- Trace Metal Services
- Material Sciences Testing
End User Insight and Forecast 2026 - 2035
- Pharmaceutical Manufacturers
- Biotech Companies
- Contract Research Organizations
- Academic & Research Institutes
Europe TIC Market for Pharmaceuticals and Biotech Industry by Region
- Germany
- By Service Type
- By Sourcing Type
- By Industry Vertical
- By End User
- U.K.
- By Service Type
- By Sourcing Type
- By Industry Vertical
- By End User
- France
- By Service Type
- By Sourcing Type
- By Industry Vertical
- By End User
- Italy
- By Service Type
- By Sourcing Type
- By Industry Vertical
- By End User
- Spain
- By Service Type
- By Sourcing Type
- By Industry Vertical
- By End User
- Russia
- By Service Type
- By Sourcing Type
- By Industry Vertical
- By End User
- Rest of Europe
- By Service Type
- By Sourcing Type
- By Industry Vertical
- By End User
Table of Contents for Europe TIC Market for Pharmaceuticals and Biotech Industry Report
1. Research Overview
1.1. The Report Offers
1.2. Market Coverage
1.2.1. By
Service Type
1.2.2. By
Sourcing Type
1.2.3. By
Industry Vertical
1.2.4. By
End User
1.3. Research Phases
1.4. Limitations
1.5. Market Methodology
1.5.1. Data Sources
1.5.1.1.
Primary Research
1.5.1.2.
Secondary Research
1.5.2. Methodology
1.5.2.1.
Data Exploration
1.5.2.2.
Forecast Parameters
1.5.2.3.
Data Validation
1.5.2.4.
Assumptions
1.5.3. Study Period & Data Reporting Unit
2. Executive Summary
3. Industry Overview
3.1. Industry Dynamics
3.1.1. Market Growth Drivers
3.1.2. Market Restraints
3.1.3. Key Market Trends
3.1.4. Major Opportunities
3.2. Industry Ecosystem
3.2.1. Porter’s Five Forces Analysis
3.2.2. Recent Development Analysis
3.2.3. Value Chain Analysis
3.3. Competitive Insight
3.3.1. Competitive Position of Industry
Players
3.3.2. Market Attractive Analysis
3.3.3. Market Share Analysis
4. Europe Market Estimate and Forecast
4.1. Europe Market Overview
4.2. Europe Market Estimate and Forecast to 2035
5. Market Segmentation Estimate and Forecast
5.1. By Service Type
5.1.1. Testing Services
5.1.1.1. Market Definition
5.1.1.2. Market Estimation and Forecast to 2035
5.1.2. Inspection Services
5.1.2.1. Market Definition
5.1.2.2. Market Estimation and Forecast to 2035
5.1.3. Certification Services
5.1.3.1. Market Definition
5.1.3.2. Market Estimation and Forecast to 2035
5.2. By Sourcing Type
5.2.1. Outsourced Services
5.2.1.1. Market Definition
5.2.1.2. Market Estimation and Forecast to 2035
5.2.2. In-house Services
5.2.2.1. Market Definition
5.2.2.2. Market Estimation and Forecast to 2035
5.3. By Industry Vertical
5.3.1. Biopharmaceutical & Biologics Analysis
5.3.1.1. Market Definition
5.3.1.2. Market Estimation and Forecast to 2035
5.3.2. Environmental Monitoring
5.3.2.1. Market Definition
5.3.2.2. Market Estimation and Forecast to 2035
5.3.3. Extractables & Leachables Studies
5.3.3.1. Market Definition
5.3.3.2. Market Estimation and Forecast to 2035
5.3.4. Sterile Compounding
5.3.4.1. Market Definition
5.3.4.2. Market Estimation and Forecast to 2035
5.3.5. Trace Metal Services
5.3.5.1. Market Definition
5.3.5.2. Market Estimation and Forecast to 2035
5.3.6. Material Sciences Testing
5.3.6.1. Market Definition
5.3.6.2. Market Estimation and Forecast to 2035
5.4. By End User
5.4.1. Pharmaceutical Manufacturers
5.4.1.1. Market Definition
5.4.1.2. Market Estimation and Forecast to 2035
5.4.2. Biotech Companies
5.4.2.1. Market Definition
5.4.2.2. Market Estimation and Forecast to 2035
5.4.3. Contract Research Organizations
5.4.3.1. Market Definition
5.4.3.2. Market Estimation and Forecast to 2035
5.4.4. Academic & Research Institutes
5.4.4.1. Market Definition
5.4.4.2. Market Estimation and Forecast to 2035
6. Germany Market Estimate and Forecast
6.1. By
Service Type
6.2. By
Sourcing Type
6.3. By
Industry Vertical
6.4. By
End User
7. U.K. Market Estimate and Forecast
7.1. By
Service Type
7.2. By
Sourcing Type
7.3. By
Industry Vertical
7.4. By
End User
8. France Market Estimate and Forecast
8.1. By
Service Type
8.2. By
Sourcing Type
8.3. By
Industry Vertical
8.4. By
End User
9. Italy Market Estimate and Forecast
9.1. By
Service Type
9.2. By
Sourcing Type
9.3. By
Industry Vertical
9.4. By
End User
10. Spain Market Estimate and Forecast
10.1. By
Service Type
10.2. By
Sourcing Type
10.3. By
Industry Vertical
10.4. By
End User
11. Russia Market Estimate and Forecast
11.1. By
Service Type
11.2. By
Sourcing Type
11.3. By
Industry Vertical
11.4. By
End User
12. Rest of Europe Market Estimate and Forecast
12.1. By
Service Type
12.2. By
Sourcing Type
12.3. By
Industry Vertical
12.4. By
End User
13. Company Profiles
13.1.
ALS Limited
13.1.1.
Snapshot
13.1.2.
Overview
13.1.3.
Offerings
13.1.4.
Financial
Insight
13.1.5.
Recent
Developments
13.2.
Applus+
13.2.1.
Snapshot
13.2.2.
Overview
13.2.3.
Offerings
13.2.4.
Financial
Insight
13.2.5.
Recent
Developments
13.3.
Bureau Veritas
13.3.1.
Snapshot
13.3.2.
Overview
13.3.3.
Offerings
13.3.4.
Financial
Insight
13.3.5.
Recent
Developments
13.4.
DEKRA SE
13.4.1.
Snapshot
13.4.2.
Overview
13.4.3.
Offerings
13.4.4.
Financial
Insight
13.4.5.
Recent
Developments
13.5.
Eurofins Scientific
13.5.1.
Snapshot
13.5.2.
Overview
13.5.3.
Offerings
13.5.4.
Financial
Insight
13.5.5.
Recent
Developments
13.6.
Intertek Group plc
13.6.1.
Snapshot
13.6.2.
Overview
13.6.3.
Offerings
13.6.4.
Financial
Insight
13.6.5.
Recent
Developments
13.7.
SGS SA
13.7.1.
Snapshot
13.7.2.
Overview
13.7.3.
Offerings
13.7.4.
Financial
Insight
13.7.5.
Recent
Developments
13.8.
The British Standards Institution
13.8.1.
Snapshot
13.8.2.
Overview
13.8.3.
Offerings
13.8.4.
Financial
Insight
13.8.5.
Recent
Developments
13.9.
TÜV SÜD
13.9.1.
Snapshot
13.9.2.
Overview
13.9.3.
Offerings
13.9.4.
Financial
Insight
13.9.5.
Recent
Developments
13.10.
UL LLC
13.10.1.
Snapshot
13.10.2.
Overview
13.10.3.
Offerings
13.10.4.
Financial
Insight
13.10.5.
Recent
Developments
14. Appendix
14.1. Exchange Rates
14.2. Abbreviations
Note: Financial insight and recent developments of different companies are subject to the availability of information in the secondary domain.
Frequently Asked Questions
Purchase Options
Latest Report
Research Methodology
- Desk Research / Pilot Interviews
- Build Market Size Model
- Research and Analysis
- Final Deliverable
Connect With Our Sales Team
- Toll-Free: +1-888-253-3960
- Phone: +91 9960 288 381
- Email: enquiry@vynzresearch.com
Europe TIC Market for Pharmaceuticals and Biotech Industry
