APAC Track and Trace Solutions Market Size & Share - Growth Forecast Report (2026-2035)
Industry Insight by Product (Hardware, Software), by Application (Serialization solutions, Aggregation solutions, Tracking, tracing, and reporting solutions), by End Use (Pharmaceutical companies, Biotechnology companies, Medical device companies, Others)
| Status : Published | Published On : May, 2026 | Report Code : VRHC1335 | Industry : Healthcare | Available Format :
|
Page : 132 |
APAC Track and Trace Solutions Market Overview
The APAC track and trace solutions market, which was valued at approximately USD 1.18 billion in 2025 and is estimated to rise to almost USD 1.45 billion by 2026, is projected to reach around USD 8.55 billion in 2035, expanding at a CAGR of about 21.9% during the forecast period from 2026 to 2035.
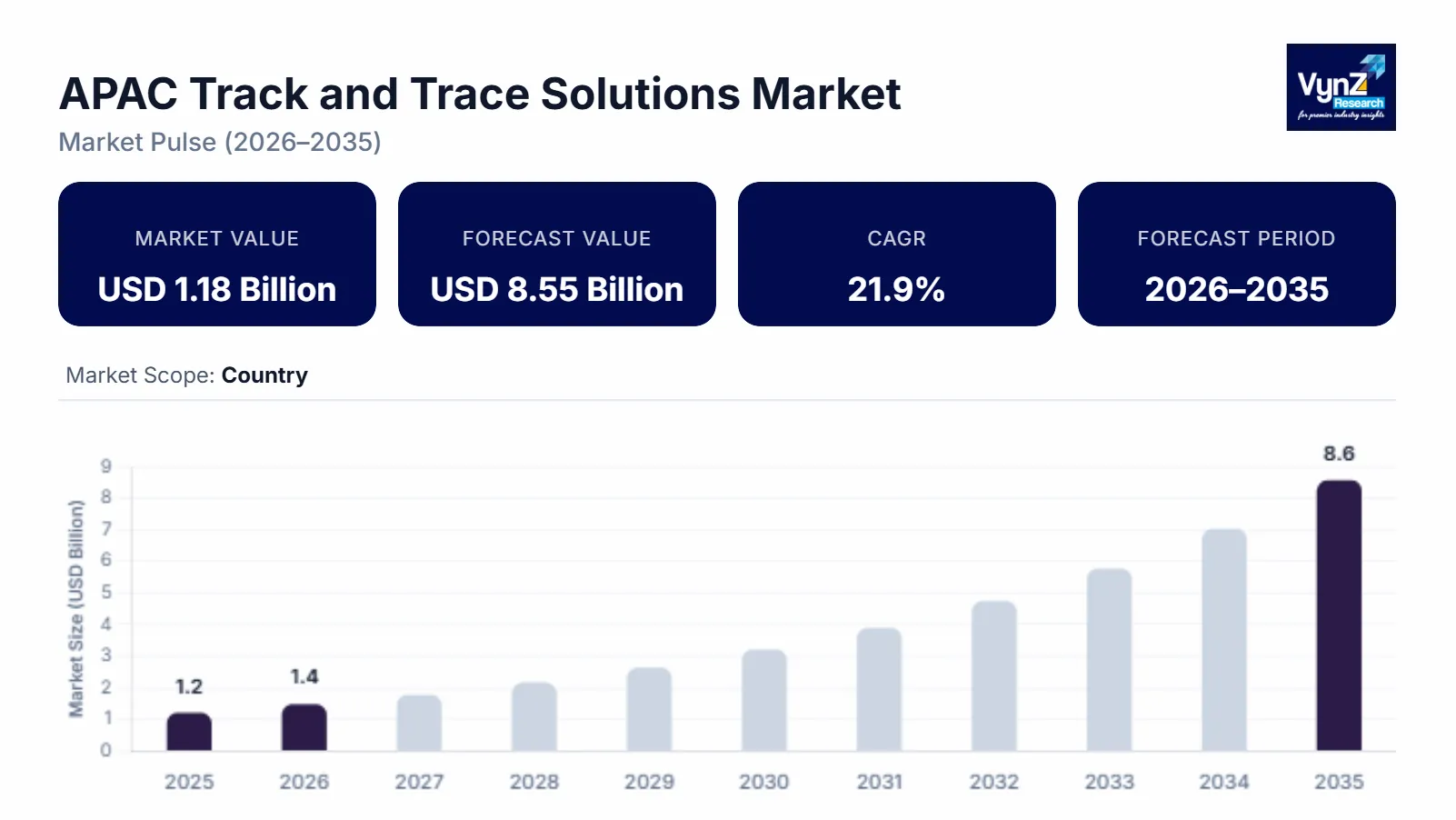
The market experiences growth because companies must comply with new product serialization rules while organizations work to prevent counterfeit drug production and they implement supply chain transparency solutions that digital track and trace systems. Global health authorities, including the World Health Organization, demonstrate a rising threat from low-quality and fake medical products which creates a continuous demand for products that enable verification and product tracking. The rising need for pharmaceutical safety monitoring together with real-time supply chain visibility and the ongoing development of regulatory compliance systems and digital healthcare systems, drives market growth in major regions of China, India and Japan.
The government programs which focus on bettering drug safety laws and establishing serialization requirements receive backing from public health organizations and national regulatory authorities, which leads to broader use of track-and-trace systems. The healthcare sector's growing emphasis on patient safety combined with their commitment to quality assurance establishes a foundation for sustained technological use throughout the regional healthcare system.
APAC Track and Trace Solutions Market Dynamics
Market Trends
Pharmaceutical and healthcare supply chains in the APAC region show technology usage and procurement patterns that undergo significant changes throughout their track and trace solutions industry. The market experiences a fundamental shift through the rising implementation of serialization and aggregation systems, which organizations use to meet their needs for compliance and transparency and operational efficiency. The World Health Organization provides guidance on combating substandard and falsified medical products, which leads to the establishment of traceability systems that operate throughout manufacturing and distribution systems. The integration of cloud-based platforms with real time monitoring solutions represents a developing trend that organizations implement to meet digital transformation requirements and regulatory standards. The market forces companies to develop integrated scalable solutions because these developments affect product offerings.
Growth Drivers
The market experiences its main growth because pharmaceutical manufacturers and distributors need to combat counterfeit drugs which results in continuous market demand. Global health authorities report increasing risks from falsified medicines, which drives the implementation of authentication technologies in healthcare systems. The market grows because healthcare organizations increase their investments in medical facilities and regulatory compliance systems. Digitalization of supply chains, which organizations use to modernize their operations, helps to increase market demand for new technologies. The demand for advanced track and trace solutions will maintain its strength during the forecast period because enterprises need traceability and operational visibility and government regulations will support their requirements.
Market Restraints / Challenges
The market encounters specific obstacles that will restrict its development, which hinders its positive growth forecast. The multiple country regulatory system creates difficulties for small and medium enterprises because it affects their ability to implement and standardize processes. The operational challenges at companies which operate in multiple countries result from their need to handle different compliance requirements and changing regulatory systems. The manufacturing and supply operations of companies face difficulties because they need advanced digital systems combined with technology services from outside partners. The requirement for companies to maintain digital system upkeep results in both financial burdens and technological system integration problems, which decrease market productivity during periods of technological progress and economic instability.
Market Opportunities
The market presents significant opportunities in the expansion of digital healthcare ecosystems, particularly driven by increasing focus on supply chain transparency and patient safety. The pharmaceutical industry and logistics providers show increasing need for traceability solutions, which enables companies to provide both adaptable and expandable solutions. Digital enabled solutions which organizations currently invest in create security and efficiency improvements through their adoption of blockchain and data analytics technologies. Global health organizations which support drug safety and quality assurance initiatives will drive larger adoption of these solutions, which will improve traceability and operational performance through their integration of advanced automation and intelligent systems.
APAC Track and Trace Solutions Market Report Coverage
|
Report Metric |
Details |
|
Historical Period |
2020 - 2024 |
|
Base Year Considered |
2025 |
|
Forecast Period |
2026 - 2035 |
|
Market Size in 2025 |
USD 1.18 Billion |
|
Revenue Forecast in 2035 |
USD 8.55 Billion |
|
Growth Rate |
21.9% |
|
Segments Covered in the Report |
Product, Application, End Use |
|
Report Scope |
Market Trends, Drivers, and Restraints; Revenue Estimation and Forecast; Segmentation Analysis; Companies’ Strategic Developments; Market Share Analysis of Key Players; Company Profiling |
|
Regions Covered in the Report |
China, India, Japan, Australia, Rest of Asia |
|
Key Companies |
ACG World, Adents, Antares Vision SpA, Axway, Dover Corp, Korber AG, Mettler-Toledo International Inc., Optimum Interactive USA Ltd, Siemens AG, TraceLink |
|
Customization |
Available upon request |
APAC Track and Trace Solutions Market Segmentation
By Product
The market for 2025 had its largest segment revenue from hardware which provided 56% of total revenues to the market. The widespread use of printing, labeling and scanning equipment in pharmaceutical manufacturing and packaging operations leads to its market dominance because it enables companies to meet serialization requirements. Production facilities continue to adopt hardware solutions because regulatory systems match worldwide health authority recommendations which focus on fighting against counterfeit medicine production. Manufacturing automation and quality assurance system investments sustain demand throughout Asian pharmaceutical hubs which specifically benefit from these systems.
The software sector will achieve the highest growth rate because its estimated annual growth rate will reach 22.6% during the period from 2026 to 2035. Companies need data management, real time monitoring and regulatory compliance solutions to deal with their complicated supply chain operations. Organizations achieve better operational performance through the increased use of cloud-based platforms and enterprise resource planning systems which enable them to monitor their business operations. The pharmaceutical and biotechnology sectors are experiencing increased adoption because government programs support digital healthcare infrastructure development and reliable data transmission.
By Application
The market in 2025 was mainly driven by serialization solutions which generated about 49% of overall segment revenue. The pharmaceutical supply chain system requires unique product identification and authentication processes because the law mandates that all products must be tracked through the supply chain. Global health organizations have emphasized serialization as a critical tool in preventing counterfeit drug circulation which continues to influence adoption across manufacturers and distributors. The region experiences increased demand for serialization technologies because pharmaceutical exports and cross border trade activities continue to grow.
Aggregation and advanced tracking solutions will experience the highest market expansion during the forecast period which will see them grow at an estimated annual growth rate of 23.1%. Companies need supply chain visibility and inventory control solutions to operate their businesses effectively. The distribution networks and packaging levels of systems build an integrated system which makes it possible to operate packaging and distribution processes more effectively. Emerging economies and developed economies expand their segment presence because logistics infrastructure investments and traceability standard regulations drive their growth.
By End User
Pharmaceutical companies accounted for the largest segment in 2025 because they generated 58% of total market revenue. The company maintains its market leadership position because of three essential factors which include regulatory requirements for product safety, authentication controls and retail distribution volume. The worldwide health authority recommends track and trace systems which lead governments to support pharmaceutical manufacturers in using these complete tracking solutions. The segment demand across Asian Pacific markets grows because drug manufacturers expand their production capacity to support international export operations.
The biotechnology and medical device sector will grow the fastest because it will achieve 21.8% annual growth from 2026 to 2035. Companies are increasing their use of traceability systems for their most sensitive and valuable products because they need protection from plastic packaging theft which protects product integrity. Advanced medical technologies and therapeutics require excellent tracking systems which ensure product quality and regulatory compliance. Emerging end user segments are experiencing increased adoption because patient safety regulations are expanding and regulatory frameworks are becoming more comprehensive.

Regional Insights
China
China held the largest portion of the market in 2025 with 32% due to its extensive pharmaceutical production facilities and its requirement to implement drug serialization according to strict regulations. The industrial hubs of Beijing, Shanghai and Shenzhen maintain their role as key demand centers which drive production and distribution operations. Increasing instances of counterfeit medicines which health authorities worldwide have documented lead to higher demand from supply chains for implementation of traceability systems.
India
India achieved about 24% of the market by 2025 because of two factors which include its fast pharmaceutical production growth and its stronger regulatory enforcement for export compliance. The manufacturing and packaging facilities in key cities of Hyderabad, Mumbai and Ahmedabad are experiencing strong technology adoption. The international health organizations report that drug counterfeit prevention has become a major focus which drives the need for authentication solutions.
Japan
Japan controlled 16% of the APAC track and trace solutions market for 2025 because its advanced healthcare facilities and high digital technology adoption enable market entry. The pharmaceutical and medical device industries which operate in Tokyo and Osaka drive the demand for their markets through their ongoing business activities. The public health authorities require all organizations to establish complete quality assurance programs which meet regulatory standards by installing advanced traceability systems.
Rest of Asia Pacific
The market for 2025 saw the rest of Asia Pacific which includes Australia, South Korea and Southeast Asian economies command an 28% market share. The regions experience growth because their pharmaceutical production capabilities continue to expand and their people become more aware of drug safety and their regulators establish compliance standards which match international norms. The international organizations fund public health initiatives to drive traceability system adoption which will enhance supply chain transparency.
Competitive Landscape / Company Insights
The market is moderately to highly competitive because global and regional companies operate their businesses through product development, price management and market share expansion. Companies are increasingly investing in research, development and digital capabilities to strengthen their market position. Regulatory frameworks aligned with guidance from the World Health Organization on preventing falsified medical products are encouraging adoption of advanced traceability systems. Government backed serialization mandates and digital compliance programs across Asia Pacific are further shaping competitive dynamics and driving continuous technological advancements.
Mini Profiles
ACG World focuses on pharmaceutical packaging and track and trace solutions, supported by strong manufacturing capabilities, global distribution networks, and cost efficient operations across emerging and developed pharmaceutical markets.
Adents operates in niche serialization and traceability segments, emphasizing software driven compliance solutions, customization capabilities, and regulatory alignment for pharmaceutical supply chains across global and regional markets.
Dover Corp leverages strategic acquisitions and diversified industrial expertise to expand market presence, offering advanced marking and coding solutions supported by strong technological capabilities and global customer reach.
Korber AG focuses on integrated supply chain and track and trace technologies, supported by strong digital capabilities, automation expertise, and established presence across pharmaceutical and industrial sectors.
Siemens AG operates in premium industrial and digital solutions segments, emphasizing advanced automation, data integration, and high-performance systems to enhance traceability and operational efficiency across complex supply chains.
Key Players
- ACG World
- Adents
- Antares Vision SpA
- Axway
- Dover Corp
- Korber AG
- Mettler-Toledo International Inc.
- Optimum Interactive USA Ltd
- Siemens AG
- TraceLink
Recent Developments
In February 2026, Antares Vision SpA expanded its serialization and inspection solutions portfolio for pharmaceutical manufacturers. The development strengthens end to end traceability and supports compliance with evolving global regulatory requirements.
In May 2025, Axway enhanced its digital integration platform to support secure data exchange in track and trace systems. The upgrade improves supply chain visibility and regulatory reporting capabilities for pharmaceutical companies.
In August 2026, Mettler-Toledo International Inc. introduced advanced inspection and coding solutions integrated with traceability systems. The launch supports quality assurance and product authentication across manufacturing and packaging operations.
In March 2025, Optimum Interactive USA Ltd expanded its traceability software offerings to improve serialization compliance for life sciences companies. The initiative focuses on enhancing real time monitoring and data management capabilities.
In January 2026, TraceLink strengthened its cloud-based track and trace network by expanding partnerships across Asia Pacific. The development enhances supply chain connectivity and supports regulatory compliance for pharmaceutical distribution networks.
APAC Track and Trace Solutions Market Coverage
Product Insight and Forecast 2026 - 2035
- Hardware
- Software
Application Insight and Forecast 2026 - 2035
- Serialization solutions
- Aggregation solutions
- Tracking
- tracing
- and reporting solutions
End Use Insight and Forecast 2026 - 2035
- Pharmaceutical companies
- Biotechnology companies
- Medical device companies
- Others
APAC Track and Trace Solutions Market by Region
- China
- By Product
- By Application
- By End Use
- Japan
- By Product
- By Application
- By End Use
- India
- By Product
- By Application
- By End Use
- South Korea
- By Product
- By Application
- By End Use
- Vietnam
- By Product
- By Application
- By End Use
- Thailand
- By Product
- By Application
- By End Use
- Malaysia
- By Product
- By Application
- By End Use
- Rest of Asia-Pacific
- By Product
- By Application
- By End Use
Table of Contents for APAC Track and Trace Solutions Market Report
1. Research Overview
1.1. The Report Offers
1.2. Market Coverage
1.2.1. By
Product
1.2.2. By
Application
1.2.3. By
End Use
1.3. Research Phases
1.4. Limitations
1.5. Market Methodology
1.5.1. Data Sources
1.5.1.1.
Primary Research
1.5.1.2.
Secondary Research
1.5.2. Methodology
1.5.2.1.
Data Exploration
1.5.2.2.
Forecast Parameters
1.5.2.3.
Data Validation
1.5.2.4.
Assumptions
1.5.3. Study Period & Data Reporting Unit
2. Executive Summary
3. Industry Overview
3.1. Industry Dynamics
3.1.1. Market Growth Drivers
3.1.2. Market Restraints
3.1.3. Key Market Trends
3.1.4. Major Opportunities
3.2. Industry Ecosystem
3.2.1. Porter’s Five Forces Analysis
3.2.2. Recent Development Analysis
3.2.3. Value Chain Analysis
3.3. Competitive Insight
3.3.1. Competitive Position of Industry
Players
3.3.2. Market Attractive Analysis
3.3.3. Market Share Analysis
4. APAC Market Estimate and Forecast
4.1. APAC Market Overview
4.2. APAC Market Estimate and Forecast to 2035
5. Market Segmentation Estimate and Forecast
5.1. By Product
5.1.1. Hardware
5.1.1.1. Market Definition
5.1.1.2. Market Estimation and Forecast to 2035
5.1.2. Software
5.1.2.1. Market Definition
5.1.2.2. Market Estimation and Forecast to 2035
5.2. By Application
5.2.1. Serialization solutions
5.2.1.1. Market Definition
5.2.1.2. Market Estimation and Forecast to 2035
5.2.2. Aggregation solutions
5.2.2.1. Market Definition
5.2.2.2. Market Estimation and Forecast to 2035
5.2.3. Tracking
5.2.3.1. Market Definition
5.2.3.2. Market Estimation and Forecast to 2035
5.2.4. tracing
5.2.4.1. Market Definition
5.2.4.2. Market Estimation and Forecast to 2035
5.2.5. and reporting solutions
5.2.5.1. Market Definition
5.2.5.2. Market Estimation and Forecast to 2035
5.3. By End Use
5.3.1. Pharmaceutical companies
5.3.1.1. Market Definition
5.3.1.2. Market Estimation and Forecast to 2035
5.3.2. Biotechnology companies
5.3.2.1. Market Definition
5.3.2.2. Market Estimation and Forecast to 2035
5.3.3. Medical device companies
5.3.3.1. Market Definition
5.3.3.2. Market Estimation and Forecast to 2035
5.3.4. Others
5.3.4.1. Market Definition
5.3.4.2. Market Estimation and Forecast to 2035
6. China Market Estimate and Forecast
6.1. By
Product
6.2. By
Application
6.3. By
End Use
7. Japan Market Estimate and Forecast
7.1. By
Product
7.2. By
Application
7.3. By
End Use
8. India Market Estimate and Forecast
8.1. By
Product
8.2. By
Application
8.3. By
End Use
9. South Korea Market Estimate and Forecast
9.1. By
Product
9.2. By
Application
9.3. By
End Use
10. Vietnam Market Estimate and Forecast
10.1. By
Product
10.2. By
Application
10.3. By
End Use
11. Thailand Market Estimate and Forecast
11.1. By
Product
11.2. By
Application
11.3. By
End Use
12. Malaysia Market Estimate and Forecast
12.1. By
Product
12.2. By
Application
12.3. By
End Use
13. Rest of Asia-Pacific Market Estimate and Forecast
13.1. By
Product
13.2. By
Application
13.3. By
End Use
14. Company Profiles
14.1.
ACG World
14.1.1.
Snapshot
14.1.2.
Overview
14.1.3.
Offerings
14.1.4.
Financial
Insight
14.1.5.
Recent
Developments
14.2.
Adents
14.2.1.
Snapshot
14.2.2.
Overview
14.2.3.
Offerings
14.2.4.
Financial
Insight
14.2.5.
Recent
Developments
14.3.
Antares Vision SpA
14.3.1.
Snapshot
14.3.2.
Overview
14.3.3.
Offerings
14.3.4.
Financial
Insight
14.3.5.
Recent
Developments
14.4.
Axway
14.4.1.
Snapshot
14.4.2.
Overview
14.4.3.
Offerings
14.4.4.
Financial
Insight
14.4.5.
Recent
Developments
14.5.
Dover Corp
14.5.1.
Snapshot
14.5.2.
Overview
14.5.3.
Offerings
14.5.4.
Financial
Insight
14.5.5.
Recent
Developments
14.6.
Korber AG
14.6.1.
Snapshot
14.6.2.
Overview
14.6.3.
Offerings
14.6.4.
Financial
Insight
14.6.5.
Recent
Developments
14.7.
Mettler-Toledo International Inc.
14.7.1.
Snapshot
14.7.2.
Overview
14.7.3.
Offerings
14.7.4.
Financial
Insight
14.7.5.
Recent
Developments
14.8.
Optimum Interactive USA Ltd
14.8.1.
Snapshot
14.8.2.
Overview
14.8.3.
Offerings
14.8.4.
Financial
Insight
14.8.5.
Recent
Developments
14.9.
Siemens AG
14.9.1.
Snapshot
14.9.2.
Overview
14.9.3.
Offerings
14.9.4.
Financial
Insight
14.9.5.
Recent
Developments
14.10.
TraceLink
14.10.1.
Snapshot
14.10.2.
Overview
14.10.3.
Offerings
14.10.4.
Financial
Insight
14.10.5.
Recent
Developments
15. Appendix
15.1. Exchange Rates
15.2. Abbreviations
Note: Financial insight and recent developments of different companies are subject to the availability of information in the secondary domain.
Purchase Options
Latest Report
Research Methodology
- Desk Research / Pilot Interviews
- Build Market Size Model
- Research and Analysis
- Final Deliverable
Connect With Our Sales Team
- Toll-Free: +1-888-253-3960
- Phone: +91 9960 288 381
- Email: enquiry@vynzresearch.com
APAC Track and Trace Solutions Market
