Global Aortic Valve Market – Analysis and Forecast (2025-2030)
Industry Insights by Product (Transcatheter Aortic Valve, Sutureless Aortic Valve, and Others), by Surgery Type (Minimally Invasive Surgery, and Open Surgery), by End User (Hospitals, and Ambulatory Surgery Centers) and by Geography (North America, Europe, Asia-Pacific, Middle East, South America and Rest of the World)
| Status : Upcoming | Report Code : VRHC1117 | Industry : Healthcare | Available Format :

|
Global Aortic Valve Market – Analysis and Forecast (2025-2030)
Industry Insights by Product (Transcatheter Aortic Valve, Sutureless Aortic Valve, and Others), by Surgery Type (Minimally Invasive Surgery, and Open Surgery), by End User (Hospitals, and Ambulatory Surgery Centers) and by Geography (North America, Europe, Asia-Pacific, Middle East, South America and Rest of the World)
Aortic Valve Market Overview
The aortic valve is a valve between the left ventricle and aorta. It divides the heart’s key pumping chamber and the aorta that provides oxygen intense blood to the body. Aortic valve disease restricts blood flow and forces the heart to work tougher to supply blood to the rest of the body. Aortic valve stenosis, aortic valve regurgitation is the aortic valve diseases. It is treated with procedures such as aortic valve repair and aortic valve replacement. These procedures help to restore normal blood flow, help to preserve the function of the heart muscles, and reduce symptoms.
The global aortic valve market is growing at a significant rate, due to the mounting occurrence of valvular diseases, and expanding programs for creating alertness about valve replacement. Different products such as a transcatheter aortic valve, sutureless aortic valve, and others contributed to the aortic valve market size. The market has witnessed high demand for transcatheter aortic valve due to a mounting number of clinical studies for enlarging their uses, mounting occurrence of cardiovascular diseases, and escalating initiatives for research activities.
Market Segmentation
Insight by Product
On the basis of product, the market is subdivided into transcatheter aortic valve, sutureless aortic valve, and others. Of all products, transcatheter aortic valve segment accounted for the larger share in the market due to a mounting number of clinical studies for enlarging their uses, mounting occurrence of cardiovascular diseases, and escalating initiatives for research activities.
Insight by Surgery Type
On the basis of surgery type, the market is subdivided into minimally invasive surgeries, and open surgery. Of both the surgery types, the open surgery accounted foremost share in the aortic valve market. Moreover, the minimally invasive surgeries are expected to grow at a faster rate in the market due to mounting acceptance of minimally invasive surgeries owing to its benefits such as faster recovery, and lesser trauma.
Insight by End User
The different end users of the market are hospitals and ambulatory surgery centers. Among all end users, the hospitals accounted for the larger share in the aortic valve market, due to a mounting number of patients in the hospital, and escalating occurrence of valvular diseases. Moreover, the ambulatory surgery centers are expected to grow at the fastest rate in the market.
Global Aortic Valve Market Report Coverage
|
Report Metric |
Details |
|
Historical Period |
2020 - 2024 |
|
Base Year Considered |
2025 |
|
Forecast Period |
2026 - 2035 |
|
Market Size in 2025 |
U.S.D. xx.x Billion |
|
Revenue Forecast in 2035 |
U.S.D. xx.x Billion |
|
Growth Rate |
x.x% |
|
Segments Covered in the Report |
By Product, By Surgery Type and By End User |
|
Report Scope |
Market Trends, Drivers, and Restraints; Revenue Estimation and Forecast; Segmentation Analysis; Companies’ Strategic Developments; Market Share Analysis of Key Players; Company Profiling |
|
Regions Covered in the Report |
North America, Europe, Asia-Pacific, Middle East, South America and Rest of the World |
Industry Dynamics
Growth Drivers
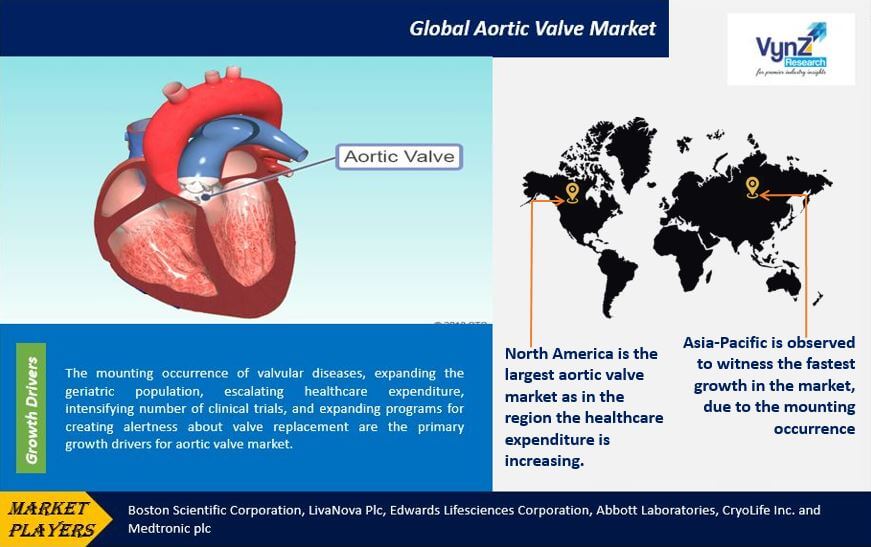
The mounting occurrence of valvular diseases, expanding the geriatric population, escalating healthcare expenditure, intensifying number of clinical trials, and expanding programs for creating alertness about valve replacement are the primary growth drivers for aortic valve market. For instance, according to the World Health Organization (WHO), globally in 2010 approximately 524 million people were aged 65 years and older and is expected to reach 1.5 billion in 2050.
Mounting inclination towards minimally invasive surgeries, escalating number of product launches and approvals, and favorable reimbursement scenario are also facilitating the growth for the aortic valve market.
Challenges
Strict regulatory requirements and product recalls are the major challenges for the growth of aortic valve market.
Industry Ecosystem
Globally, the manufacturing companies trying to enter the aortic valve market are required to maintain stringent regulatory standards. Moreover, the high level of capital requirement also poses a major barrier to the entry of new players. This offers an edge to the established players in the industry competition.
Geographic Overview
Geographically, North America is the largest aortic valve market as in the region the healthcare expenditure is increasing. In addition, the mounting occurrence of valvular diseases, expanding the geriatric population, the presence of a large number of medical device manufacturers, and advanced reimbursement structure is also up surging growth of the North American aortic valve market.
Asia-Pacific is observed to witness the fastest growth in the market, due to the mounting occurrence of valvular diseases and the presence of a large number of the patient base with cardiovascular diseases. In addition, mounting healthcare expenditure, expanding geriatric population, mounting medical tourism, refining healthcare infrastructure, and escalating inclination towards minimally invasive surgery are also creating a positive impact on the aortic valve market growth in the region.
Competitive Insight
Key players in the aortic valve market are catering to the demand of these devices by investing in technologically advanced products in their product portfolio across the globe. In June 2017, Edwards Lifesciences Corporation received FDA approval for aortic and mitral valve-in-valve procedures utilizing the Edwards SAPIEN 3 transcatheter heart valve. Boston Scientific Corporation, LivaNova Plc, Edwards Lifesciences Corporation, Abbott Laboratories, CryoLife Inc. and Medtronic plc are the key players offering aortic valve.
The Aortic Valve Market report offers a comprehensive market segmentation analysis along with an estimation for the forecast period 2025–2030.
Segments Covered in the Report
- By Product
- Transcatheter aortic valve
- Sutureless aortic valve
- Others
- By Surgery Type
- Minimally invasive surgeries
- Open surgery
- By End User
- Hospitals
- Ambulatory surgery centers
Geographical Segmentation
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- U.K.
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia-Pacific (APAC)
- China
- Japan
- India
- South Korea
- Rest of Asia-Pacific
- Rest of the World (RoW)
- Brazil
- Saudi Arabia
- South Africa
- U.A.E.
- Other Countries
Frequently Asked Questions
Purchase Options
Latest Report
Research Methodology
- Desk Research / Pilot Interviews
- Build Market Size Model
- Research and Analysis
- Final Deliverable
Connect With Our Sales Team
- Toll-Free: 1 888 253 3960
- Phone: +91 9960 288 381
- Email: enquiry@vynzresearch.com
Aortic Valve Market
