Surgical Site Infection Prevention Market – Analysis and Forecast (2026-2035)
Industry Insights by Product (Wound Care Dressings, Antibiotic Prophylaxis, Clippers, Surgical Drapes, Antiseptics and Disinfectants, Surgical Staff Clothing, Patient-Warming Products, Wound Irrigation Products, Wound Care Supplementary Products and Others), by Phases (Postoperative, Preoperative and Intraoperative), and by End User (Ambulatory Surgery Centers, Hospitals, Clinics and Others), and by Geography (U.S., Canada, Germany, U.K., France, China, Japan, India, and Rest of the World)
| Status : Published | Published On : Jan, 2026 | Report Code : VRHC1045 | Industry : Healthcare | Available Format :

|
Page : 250 |
Surgical Site Infection Prevention Market – Analysis and Forecast (2026-2035)
Industry Insights by Product (Wound Care Dressings, Antibiotic Prophylaxis, Clippers, Surgical Drapes, Antiseptics and Disinfectants, Surgical Staff Clothing, Patient-Warming Products, Wound Irrigation Products, Wound Care Supplementary Products and Others), by Phases (Postoperative, Preoperative and Intraoperative), and by End User (Ambulatory Surgery Centers, Hospitals, Clinics and Others), and by Geography (U.S., Canada, Germany, U.K., France, China, Japan, India, and Rest of the World)
Surgical Site Infection Prevention Market Overview
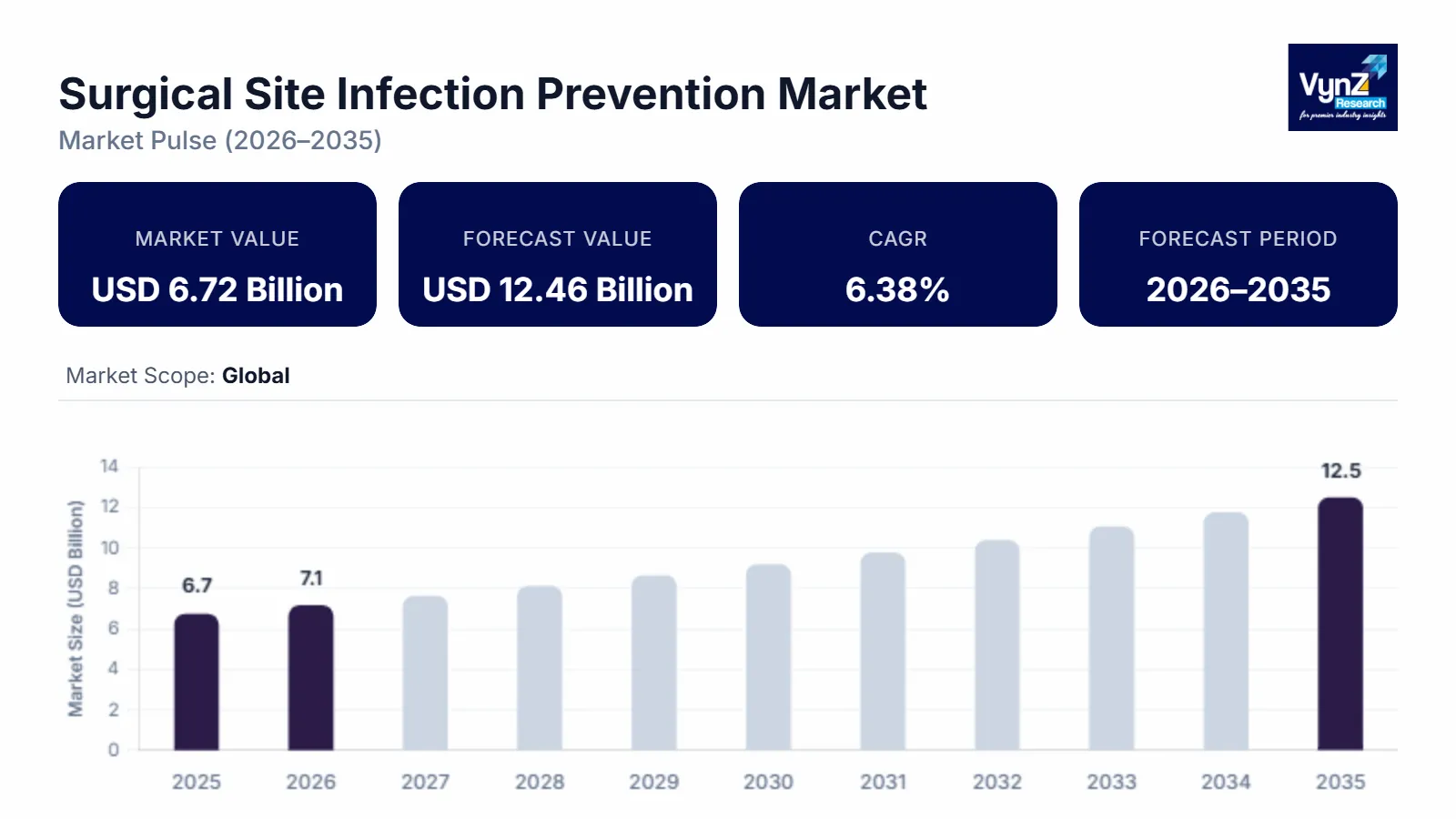
The surgical site infection prevention market refers to the market for products and solutions aimed at reducing the occurrence of surgical site infections (SSIs) in patients undergoing surgical procedures. Surgical site infections are a significant concern in healthcare settings as they can lead to prolonged hospital stays, increased healthcare costs, and patient morbidity and mortality. Various factors contribute to the development of surgical site infections, including bacterial colonization of the surgical site, compromised patient immune systems, improper sterile techniques, and inadequate post-operative wound care. To address these challenges, several companies and healthcare organizations offer a range of products and solutions designed to prevent surgical site infections. The global surgical site infection prevention market was valued at USD 6.72 billion in 2025 and is estimated to reach around USD 7.1 billion in 2026. It is projected to reach around USD 12.46 billion by 2035, expanding at a CAGR of about 6.38 % during the forecast period from 2026 to 2035. Surgical site infection is a septicity that arises after surgery, which can include the skin or tissues, organs, or implanted material. The global surgical site infection prevention market is growing at a significant rate, due to the mounting occurrence of surgical site infections and the innovation of technologically advanced products. Different products contributed to the surgical site infection prevention market size. The market has witnessed high demand for antibiotic prophylaxis over the last few years, as it is a preventive measure that healthcare facilities implement to prevent infection and contamination throughout the treatment.
Surgical Site Infection Prevention Market Segmentation
Insight by Product
- wound care dressings
- antibiotic prophylaxis products
- clippers
- surgical drapes
- antiseptics and disinfectants
- surgical staff clothing
- patient-warming products
- wound irrigation products
- wound care supplementary products
- others
On the basis of product, the surgical site infection prevention market is subdivided into wound care dressings, antibiotic prophylaxis products, clippers, surgical drapes, antiseptics and disinfectants, surgical staff clothing, patient-warming products, wound irrigation products, wound care supplementary products and others. Of all products, antibiotic prophylaxis products accounted largest share in the market as it is the most popular type of preventive measure that healthcare facilities implement to prevent infection and contamination throughout the treatment. In addition, antiseptics and disinfectants are expected to grow at fastest rate in the market.
Insight by Phases
- postoperative phase
- preoperative phase
- intraoperative phase
On the basis of phase the market is subdivided into postoperative phase, preoperative phase and intraoperative phase. Elevated quantity of products exhausted to mitigate the possibilities of infection and adherence of healthcare professionals to infection-control rules are some of the factors responsible for the growth of the intraoperative phase. Of all the phase, the intraoperative phase accounted foremost share in the surgical site infection prevention market.
Insight by End User
- ambulatory surgery centers
- hospitals
- clinics
- others
The different end-users of the market are ambulatory surgery centers, hospitals, clinics and others. Mounting inclination towards ambulatory surgery centers owing to boosting privacy during the stay is the factor growing the demand of surgical site infection prevention in ambulatory surgery centers. Among all end-users, ambulatory surgery centers are expected to grow at fastest rate in the surgical site infection prevention market.
Global Surgical Site Infection Prevention Market Report Coverage
|
Report Metric |
Details |
|
Historical Period |
2020 - 2024 |
|
Base Year Considered |
2025 |
|
Forecast Period |
2026 - 2035 |
|
Market Size in 2025 |
U.S.D. 6.72 Billion |
|
Revenue Forecast in 2035 |
U.S.D. 12.4 Billion |
|
Growth Rate |
6.38% |
|
Segments Covered in the Report |
By Product, By Phases and By End User |
|
Report Scope |
Market Trends, Drivers, and Restraints; Revenue Estimation and Forecast; Segmentation Analysis; Companies’ Strategic Developments; Market Share Analysis of Key Players; Company Profiling |
|
Regions Covered in the Report |
North America, Europe, Asia-Pacific, Middle East, and Rest of the World |
Industry Dynamics
Surgical Site Infection Prevention Market Growth Drivers
The innovation of technologically advanced products, mounting hospital-acquired infection, expanding regulatory guidelines for hospital infection prevention, and the mounting number of surgical procedures are the primary growth drivers for the surgical site infection prevention market. For instance, according to the American Society of Plastic Surgeons in 2015, approximately 15.9 million cosmetic procedures were performed in the U.S., a two percent increase over 2014.
The mounting occurrence of surgical site infections is also driving the growth of the market. For instance, according to the World Health Organization (WHO), approximately 11 percent of patients who undergo surgery are infected in the procedure in low and middle-income countries.
Mounting initiatives by several organizations, mounting healthcare expenditure, and expanding geriatric population are also facilitating the growth of the surgical site infection prevention market. For instance, according to WHO, globally in 2010 approximately 524 million people were aged 65 years and older, and is expected to reach 1.5 billion in 2050.
Surgical Site Infection Prevention Market Challenges
The less preventive methods for nosocomial infections in developing countries, a lack of alertness about surgical site infection and lack of alertness of hospital-acquired infection prevention and control are the major challenges for the growth of the surgical site infection prevention market.
Surgical Site Infection Prevention Market Industry Ecosystem
Globally, the manufacturing companies trying to enter the surgical site infection prevention market are required to maintain stringent regulatory standards. Moreover, the high level of capital requirement also poses a major barrier for the entry of new players. This offers an edge to the established players in the industry competition.
Antimicrobial products: This includes antimicrobial sutures, dressings, and coatings that are designed to inhibit the growth of bacteria and prevent contamination at the surgical site.
Surgical drapes and gowns: These are sterile barriers used to cover the patient and maintain a sterile field during surgery, reducing the risk of pathogens reaching the incision site.
Preoperative skin preparation: Products such as antiseptic solutions, including chlorhexidine gluconate or povidone-iodine, are used to clean the patient's skin before surgery, reducing the microbial load at the site.
Sterilization and disinfection products: These products ensure that surgical instruments and equipment are properly sterilized or disinfected, minimizing the risk of introducing pathogens during the surgical procedure.
Education and training: Healthcare providers receive training and education on infection prevention protocols, including proper hand hygiene, aseptic techniques, and the use of appropriate protective equipment.
Surveillance and monitoring systems: These systems help track and monitor SSIs to identify trends, implement targeted interventions, and improve overall infection prevention practices.
Surgical Site Infection Prevention Market Geographic Overview
Geographically, North America is the largest surgical site infection prevention market as in the region the number of surgical procedure is increasing. Expanding geriatric population, technological advancement of advanced products, are also up surging growth of the North American surgical site infection prevention market. For instance, according to the U.S. Census Bureau, in 2012 approximately 43.1 million people were aged 65 years and older in the U.S., and it is expected to reach 83.7 million in 2050.
Asia-Pacific is observed to witness the fastest growth in the market, as the region comprises a large population base. In addition, mounting healthcare expenditure, the mounting number of surgical procedures, expanding the geriatric population, the mounting occurrence of surgical site infection, and refining healthcare infrastructure are also creating a positive impact on the surgical site infection prevention market growth in the region.
Surgical Site Infection Prevention Market Competitive Insight
Key players in the surgical site infection prevention market are catering to the demand for these devices by investing in technologically advanced products in their product portfolio across the globe.
Mölnlycke Health Care is a Swedish medical device company headquartered in Gothenburg. The company manufactures and sells wound care and single-use surgical products and is a service provider to the healthcare sector.
Covalon Technologies Ltd. has developed therapeutic biomaterials for wound care, surgical applications, and coatings. The Company offers covalent processes to apply hydrophilic and lubricious, durable coatings to polymeric medical devices. Covalon provides covalently cross-linked denatured collagen platforms for applications in wound care and drug delivery.
3M Company, Medtronic plc, Mölnlycke Health Care AB, B. Braun Melsungen AG, Covalon Technologies Ltd., Stryker Corporation, Becton, Dickinson and Company, Prescient Surgical Inc., Surgical Site Infection Prevention LLC, and STERIS plc are the key players offering surgical site infection prevention.
Recent Developments by Key Players
Stryker, a global leader in medical technologies, has completed the acquisition of mfPHD, a leader in modular stainless steel wall systems for hospitals and ambulatory surgery centers. Stryker shall provide Fortress, a modular wall system that consolidates construction timelines, allowing for earlier operating room utilization as well as expedited room repair when disruptive events occur. This acquisition shall allow for innovative product development opportunities to build out an ecosystem of embedded technologies delivering data and analytics for an intelligent operating room, minimizing downtime and costly renovations.
Becton, Dickinson, and Company's Dubai branch and the Emirates Nursing Association (ENA) have collaborated to focus on advancing patient safety and healthcare worker protection across the UAE. This partnership shall see to the critical areas such as complications reduction, medication management, infection prevention, and ensuring the highest standards in vascular access management.
The Surgical Site Infection Prevention Market report offers a comprehensive market segmentation analysis along with an estimation for the forecast period 2026–2035.
Segments Covered in the Report
- By Product
- Wound Care Dressings
- Antibiotic Prophylaxis
- Clippers
- Surgical Drapes
- Antiseptics and Disinfectants
- Surgical Staff Clothing
- Patient-Warming Products
- Wound Irrigation Products
- Wound Care Supplementary Products
- Others
- By Phases
- Postoperative
- Preoperative
- Intraoperative
- By End User
- Ambulatory Surgery Centers
- Hospitals
- Clinics
- Others
Region Covered in the Report
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- U.K.
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia-Pacific (APAC)
- China
- Japan
- India
- South Korea
- Rest of Asia-Pacific
- Rest of the World (RoW)
- Brazil
- Saudi Arabia
- South Africa
- U.A.E.
- Other Countries
.png)
Source: VynZ Research
Frequently Asked Questions
Purchase Options
Latest Report
Research Methodology
- Desk Research / Pilot Interviews
- Build Market Size Model
- Research and Analysis
- Final Deliverable
Connect With Our Sales Team
- Toll-Free: 1 888 253 3960
- Phone: +91 9960 288 381
- Email: enquiry@vynzresearch.com
Surgical Site Infection Prevention Market
