TIC Market for Pharmaceuticals & Biotech Industry Overview
The global TIC Market for Pharmaceuticals & Biotech Industry, which was valued at approximately USD 5.83 billion in 2025 and is estimated to reach around USD 6.07 billion in 2026, is projected to reach approximately USD 8.79 billion by 2035, expanding at a CAGR of about 4.2% during the forecast period from 2026 to 2035.
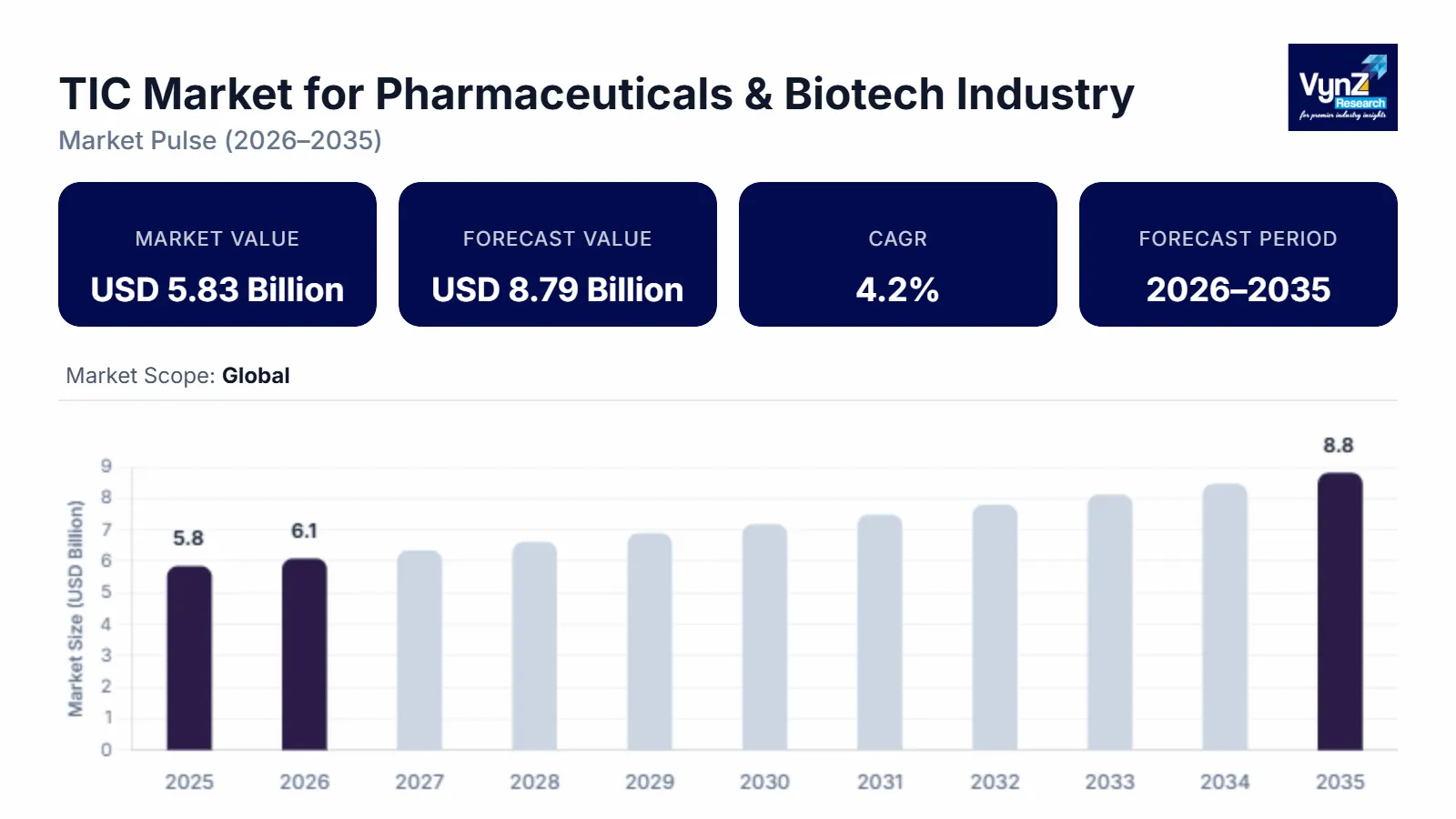
The primary factors propelling this industry are stricter regulations, the growing complexity of pharmaceutical products, especially biologics and customized drugs, and the globalization of supply chains. From its estimated USD 6.5 billion in 2025 to 2030, the pharmaceutical TIC market is expected to develop at a compound annual growth rate (CAGR) of 3–4%. Because of increased pharmaceutical output and tighter regulations, the Asia-Pacific region is rapidly growing. The digitization of testing procedures, remote audits, the application of AI-driven analytics, and the growing need for all-inclusive compliance solutions are some of the primary market trends.
TIC services are essential for product approval and marketing since regulatory agencies want thorough documentation and strict evaluation.
All things considered, the TIC market is essential to guaranteeing patient safety, preserving product integrity, and helping pharmaceutical firms comply with changing international regulatory requirements.
TIC Market for Pharmaceuticals & Biotech Industry Dynamics
Market Trends
The key concepts include the digitization of testing procedures, remote audits and the application of AI-driven analytics, along with the growing need for end-to-end compliance solutions.
Stricter global regulatory frameworks are too driving the need for third-party testing, inspection, and certification services to ensure compliance and patient safety.
Automation and artificial intelligence are transforming TIC services by improving testing accuracy, predictive analytics, and inspection efficiency. AI is being utilized more and more in drug development and quality control to improve compliance procedures while cutting costs and schedules.
Growth Drivers
Tough rules from authorities push demand for testing in pharma and biotech fields. Groups like the US FDA, EMA, or WHO require regular checks, safety tests, unless certification is confirmed. Spending on inspections tied to compliance takes up about 35–40% of quality funds - while audit rates climb roughly 6–7% each year.
Faster progress in biologics, plus similar treatments, is pushing up need for TIC. More than four out of ten drugs worldwide now fall under biologics - these involve tricky checks, approval steps, besides tight germ control. Because these medicines demand stricter safety and purity rules, testing labs are growing around 8 to 10% each year.
Fewer drugs get made where they're sold, pushing companies to check quality more often. Over 60% of key drug parts come from abroad, so inspections between countries are getting common. Because of this shift, testing each production run matters way more these days. Services that verify supply routes expand by about 7.5% every year. The primary trends include the digitization of testing procedures, remote audits, the incorporation of AI-driven analytics, and the growing need for end-to-end compliance solutions.
All things considered, the TIC market is essential to guaranteeing patient safety, preserving product integrity, and helping pharmaceutical firms comply with changing international regulatory requirements.
Market Restraints / Challenges
The testing, inspection, and certification market face certain challenges like trade wars and growth fluctuations, huge investment for automation and installation of industrial safety systems, high cost of TIC owing to diverse standards and regulations globally. Moreover, ensuring people and site safety, processes need to be optimized from extraction which requires higher pressures and temperatures, a statutory inspection of production assets, and asset life extension are hampering the oil & gas industry. Furthermore, a lack of testing facilities and skilled personnel may hamper the growth of the TIC market.
Market Opportunities
The growing need for biologics, biosimilars, and customized medications which call for sophisticated testing and certification creates significant growth prospects for the TIC market for pharmaceuticals and biotech. TIC service outsourcing is increasing due to expansion in emerging countries, particularly in Asia-Pacific. The need for end-to-end, specialist TIC solutions is further fuelled by expanding regulatory requirements and the complexity of the global supply chain. Furthermore, productivity and scalability are improved via digitalization, AI-driven testing, and remote inspections.
Global TIC Market for Pharmaceuticals & Biotech Industry Report Coverage
|
Report Metric
|
Details
|
|
Historical Period
|
2020 - 2024
|
|
Base Year Considered
|
2025
|
|
Forecast Period
|
2026 - 2035
|
|
Market Size in 2025
|
U.S.D. 5.83 Billion
|
|
Revenue Forecast in 2035
|
U.S.D. 8.79 Billion
|
|
Growth Rate
|
4.2%
|
|
Segments Covered in the Report
|
Service Type, Product Type, Stage of Manufacturing and End User
|
|
Report Scope
|
Market Trends, Drivers, and Restraints; Revenue Estimation and Forecast; Segmentation Analysis; Companies’ Strategic Developments; Market Share Analysis of Key Players; Company Profiling
|
|
Regions Covered in the Report
|
North America, Europe, Asia Pacific
|
TIC Market for Pharmaceuticals & Biotech Industry Segmentation
By Service Type
By kind of service, you’ve got testing, checks, proofing, reviews, plus lab confirmation. Testing’s growing quickest - about 9.3% each year - thanks to more need for germ tests, shelf-life analysis, and safety checks in meds and biological products.
Testing makes up about 48–50% of all TIC income since each batch must pass safety and quality checks. Product complexity keeps rising, which boosts demand for these services.
Inspection work’s picking up since officials are checking sites more often. Certifying stuff helps firms stick to production rules, whereas checkups still matter when picking suppliers or prepping for rule reviews.
By Product Type
By kind, it includes meds, biological drugs, copycat biologics, shots that prevent illness, or treatments using cells and genes. Biological medicines along with high-tech therapies grow quickest - close to 10.5% each year - because making them is tough, also rules around them are strict.
Old-school meds keep needing regular lab checks, so TIC stays steady. Vaccines need super-close monitoring now - way more since the pandemic hit.
Cell and gene treatments need unique checks - like purity, strength, or detecting unwanted elements - which boosts demand for premium testing work.
By Stage of Manufacturing
At the production phase, TIC checks happen when testing raw supplies, monitoring quality mid-way through making items, inspecting completed goods, also watching performance after launch. Testing final products expands quickest - about 9.8% each year - because tight rules demand verification before things hit stores.
Raw materials plus suppliers need checking - global sources can bring more contamination risks. During manufacturing, tests help keep things steady while confirming each batch is good.
Post-market monitoring keeps rising since officials now focus more on drug safety, while pushing clear tracking from start to finish.
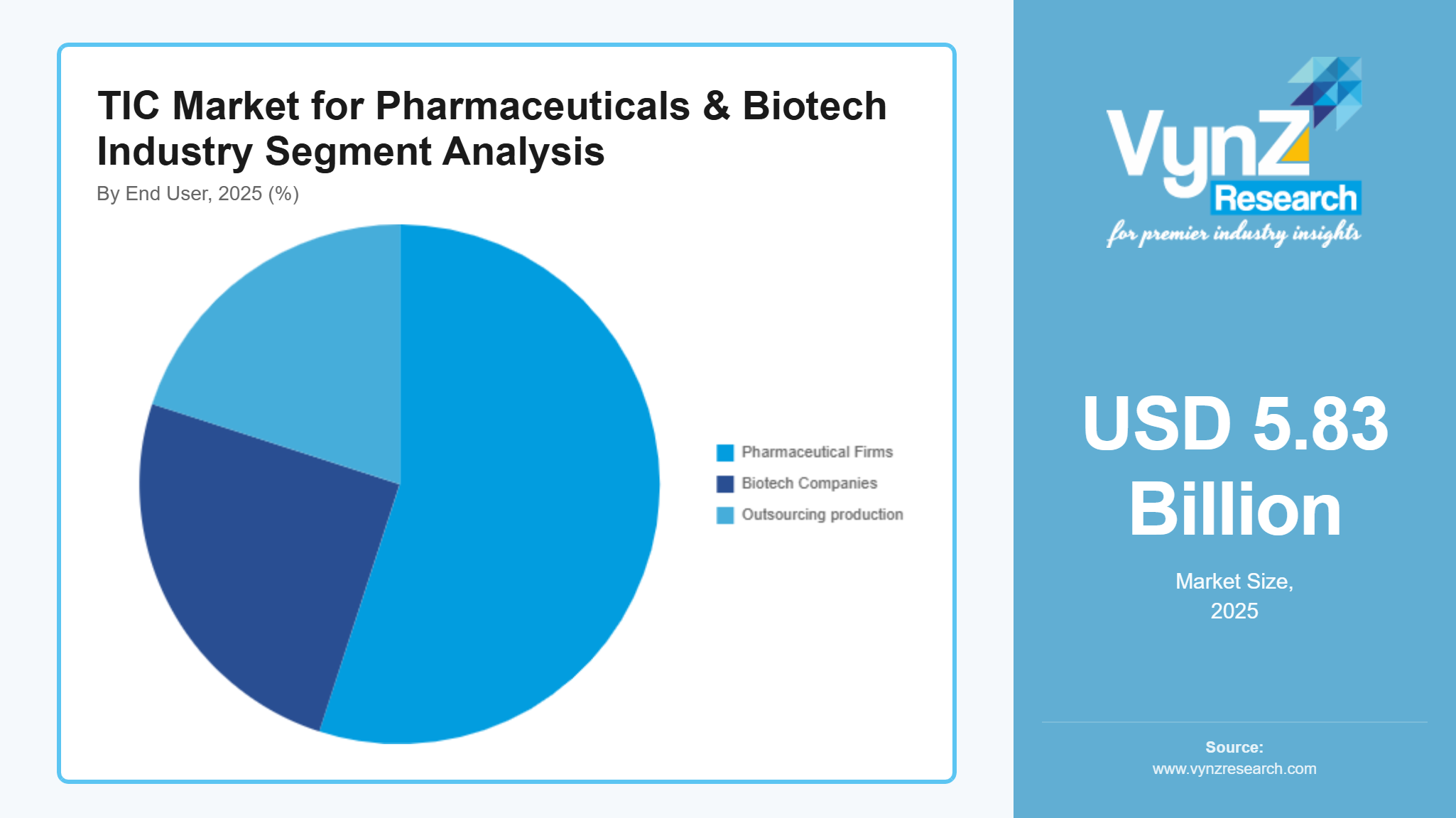
By End User
At the end-user level, you’ve got pharma makers, biotech firms, outsourcing production groups, also labs doing studies. Pharma producers lead in need - they cover around 52 to 55 percent of all testing, inspecting, certifying costs - mainly because rules keep pushing them to check everything, nonstop.
Biotech firms are growing quicker than any other users - rising by about 9.7% each year - since new ideas spread across countries. While labs push fresh breakthroughs, demand climbs steadily behind the scenes.
Contract manufacturers lean on TIC services to meet rules across various clients. Meanwhile, research labs turn to TIC when running clinical trials or small-scale production runs.
Regional Insights
North America
North America leads as the top regional market, expanding around 6.5–7% each year. In the U.S., strict FDA rules combined with large-scale biologics output drive most of the need.
FDA checks are getting tougher, which pushes up the need for review services. On top of that, new types of treatments drive more demand for niche testing and verification work.
The area's ahead in using digital tools for quality checks - this boosts performance while growing premium testing deals.
Asia Pacific
Asia Pacific's growth tops all regions - around 9 to 10% each year - thanks to rising drug production in places like China, India, and South Korea. Because this area makes more than half of the world’s active ingredients, companies there face heavy checks on suppliers, so verification needs keep climbing.
Rising exports to strict markets mean firms must meet U.S. rules - so more audits pop up. On top of that, public funding for biotech pushes testing needs higher.
Firms from abroad boost area labs while building more check points nearby.
Europe
Europe’s economy inches forward each year - around 5.8 to 6.2% - thanks to strict rules backed by the EMA and local agencies. Because biologic drugs are widely used, labs keep busy running complex tests.
Quality checks plus cert work are rising because firms follow new EU medicine rules. Green goals together with tracking laws mean more hurdles to clear.
European TIC companies target tough tests as well as rules advice, helping them stay top-tier.
Competitive Landscape / Company Insights
The largest global firms own a sizable portion of the moderately concentrated TIC industry, but there is still space for regional and specialty experts. Price is not the only factor driving competition; competencies and compliance standards play a major role. As a barrier to entrance, accreditation benefits well-established companies with solid regulatory histories. SGS SA, Bureau Veritas, Intertek Group plc, Eurofins Scientific, TÜV SÜD, and TÜV Rheinland are some of the major organizations that compete on the basis of their global reach, technological know-how, and regulatory reputation.
Mini Profiles
SGS SA, is a global leader in TIC services, SGS offers extensive testing, inspection, and certification solutions across life sciences. The company focuses on sustainability, digital compliance, and advanced analytical testing, strengthening its leadership through strategic acquisitions and innovation.
Bureau Veritas is a France-based multinational operating in over 140 countries, providing inspection, certification, and laboratory testing services. It emphasizes R&D, digital solutions, and global expansion to maintain competitiveness in regulated industries like pharmaceuticals.
Intertek Group plc is a UK-based TIC provider with a strong global laboratory network. It specializes in quality assurance, high-throughput testing, and regulatory compliance, leveraging digital tools and acquisitions to expand its healthcare and life sciences capabilities.
Eurofins Scientific is a leading laboratory testing company, Eurofins is highly focused on pharmaceutical and biotech testing, including bioanalytical services and clinical testing. Its strength lies in specialized lab networks and strong presence in life sciences.
Key Players
- Intertek Group Plc
- Bureau Veritas
- MISTRAS Group
- SGS SA
- Eurofins Scientific
- TUV Rheinland
- TUV SUD
- DEKRA SE
- Applus+
- DNV GL
Recent Developments
In March 2026, Panasonic has launched a liquid cooling systems business for AI data centers. This business focus on cooling systems for high-performance computing & generative AI servers and development of high-capacity cooling units (1,200 kW+) which aligns with rising demand from data centers and cloud computing.
In March 2026, ABB Robotics Partners with NVIDIA to Deliver Industrial-Grade Physical AI at Scale. The collaboration focuses on combining ABB Robotics’ software programming, design and simulation suite, RobotStudio, with the physically accurate simulation power of NVIDIA Omniverse libraries to close technology's long-standing 'sim-to-real’ gap. Developers can simulate robots in digital twins and generate synthetic data to train their physical AI models, enabling businesses of all types and sizes to deploy AI-driven robotics for various industrial workflows.
In February 2026, Google has awarded Form Energy, Inc. a USD billion contract to provide its cutting-edge iron-air batteries for a Minnesota data center project. One of the biggest and longest-lasting battery installations in the world, this project entails the deployment of a massive energy storage system that can provide electricity constantly for up to 100 hours. Iron-air batteries are less expensive for widespread use since they are made of inexpensive materials like iron.



