Guidewires Market Overview
Guidewires is a flexible wire inserted into a confined space to guide an instrument. It is mainly used in vascular intervention. The Global Guidewires Market is growing at a significant rate, due to escalating acceptance of minimally invasive surgery and increasing healthcare expenditure. The market has witnessed high demand for surgical guidewires over the last few years due to mounting regulatory directives executed for the prevention of healthcare-related infections and an escalating number of surgical treatments.
Guidewires imply the flexible wire that is inserted into a confined space to guide instruments and help in the proper placement of specific medical devices such as stents and catheters. It is primarily used in vascular intervention in the healthcare sector and is usually flexible and thin in design. These wires are also normally coated to allow smooth movement through vessels and arteries. The development of products ensures minimal discomfort to the patients while maximizing procedural accuracy during medical interventions.
The global guidewires market is growing at a significant rate due to the higher adoption of minimally invasive surgery. It is also attributed to the growing healthcare expenditure as well as the rising regulatory directives for promoting the prevention of healthcare-related infection. In addition, the rise in the number of surgical treatments is also propelling the market growth.
The huge expense of surgical guidewires is the most significant factor that is hindering the growth of the global guidewires market. However, rapid technological advancements have created immense opportunities for manufacturers to leverage the market potential.
Market Segmentation
Insight by Material
The global guidewires market is segmented by material into hybrid guidewire, nitinol guidewire, and stainless-steel guidewire out of which the nitinol guidewire segment is expected to hold a larger market share during the forecast period due to the growing adoption of minimally invasive surgeries and applications of nitinol guidewires in a large number of medical procedures.
Insight by Type
The global guidewires market is segmented by types into surgical and diagnostic guidewires segments. Out of these two segments, the surgical guidewires segment is expected to hold a larger share and grow at a higher CAGR during the forecast period due to the growing number of surgical treatments for neurovascular, coronary, and peripheral diseases as well as the convenient reimbursement policies for vascular treatment procedures. It is also attributed to the growing regulatory directives implemented in developed countries to prevent healthcare-associated infections.
Insight by Application
The global guidewires market is segmented based on application into vascular, cardiology, neurology, urology, oncology, gastroenterology, and otolaryngology segments. Out of all these segmentations, the cardiology segment is expected to account for the largest share of the market during the forecast period due to the rising aging population and growing number of cardiovascular diseases. It is also facilitated by the higher adoption and use of guidewires by the cardiologists for technical and procedural assistance offered especially by the nitinol materials.
Insight by End-User
The global guidewires market is segmented by the varied end-users into research laboratories & academic institutes, hospitals, diagnostic centers, surgical centers, and ambulatory care centers. Out of all these segmentations, the hospitals, diagnostic centers, and surgical centers categories will grow at a higher CAGR during the forecast period due to greater accessibility of healthcare reimbursement for minimally invasive procedures, rapid technological developments in the field of image-guided medical procedures, and growing incidents of target diseases.
Global Guidewires Market Report Coverage
|
Report Metric
|
Details
|
|
Historical Period
|
2020 - 2024
|
|
Base Year Considered
|
2025
|
|
Forecast Period
|
2026 - 2035
|
|
Market Size in 2025
|
U.S.D. xx.x Billion
|
|
Revenue Forecast in 2035
|
U.S.D. xx.x Billion
|
|
Growth Rate
|
x.x%
|
|
Segments Covered in the Report
|
By Material, By Type, By Application and By End-User
|
|
Report Scope
|
Market Trends, Drivers, and Restraints; Revenue Estimation and Forecast; Segmentation Analysis; Companies’ Strategic Developments; Market Share Analysis of Key Players; Company Profiling
|
|
Regions Covered in the Report
|
North America, Europe, Asia-Pacific, Middle East, South America and Rest of the World
|
Industry Dynamics
Industry Trends
The most notable trends within the guidewires market include the significant healthcare association of healthcare providers and an alliance of distribution competencies of major original equipment manufacturers. In addition, significant technological developments are helping produce advanced guidewires that have higher application and are customizable according to specific requirements and operated remotely. There is also a growing integration of guidewires into imaging modalities and surgical robots noticed within the market along with notable advancement in engineering that minimizes risks.
Growth Drivers
The primary factor that influences the growth of the global guidewires market is the rise in the occurrence of urological disorders, cardiovascular diseases, and Peripheral Artery Disease (PAD). In addition, the significant rise in the number of minimally invasive surgeries as well as the rising senior population where ischemic heart disease, vascular dementia, and multiple sclerosis are widespread further drives the market growth. The growth of the market is further attributed to the growing adoption and application of guidewires for treating the rising target diseases and lifestyle associated diseases. Moreover, continuous efforts by major players to launch new products, favorable reimbursement policies in developed countries, a rise in healthcare expenditure, and an increase in government initiatives in developed countries to restrict healthcare-related issues such as infection are also fueling the growth of the guidewires market.
Challenges
One significant factor that prevents wide adoption and extensive use of guidewires and expansion of the market is the shortage of trained professionals to perform minimally invasive procedures. It is also attributed to the additional cost of high excise duties for the products which makes them unaffordable for many and prevents market expansion.
Opportunities
Growth opportunities are however presented by the manufacturing companies who are investing more money in research and development of products to meet the rising demand for different medical procedures. Moreover, the strict regulatory requirements that the manufacturing companies need to abide by also present novel growth opportunities.
Geographic Overview
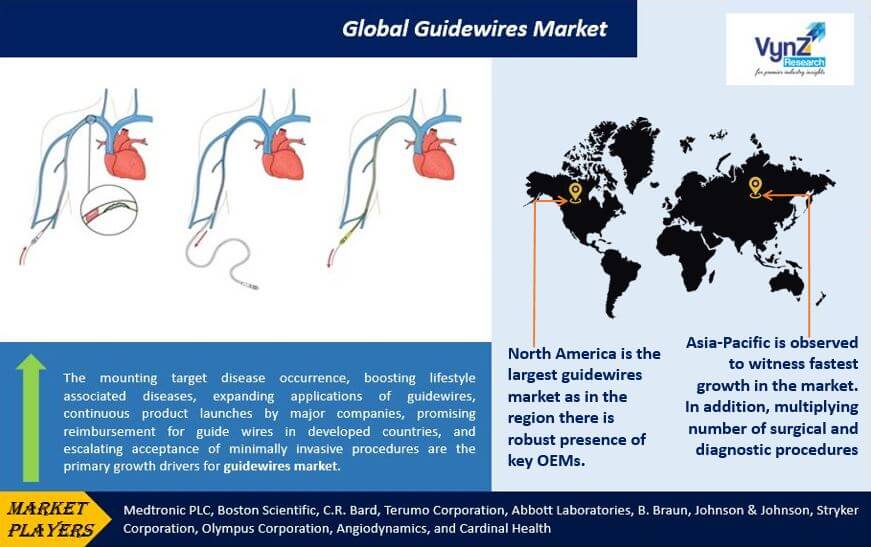
North America contributes a large share of the guidewires market due to the higher presence of major OEMs in the region. In addition, the region also has a huge target patient population base needing minimally invasive surgical procedures and a growing aging population that further propels the market growth. Apart from that, the market growth is attributed to the growing number of lifestyle-related diseases, higher acceptance of more sophisticated wires, a rising number of cardiovascular diseases, and supportive reimbursement policies.
The Asia-Pacific market will grow at a faster rate due to growing demand and number of surgical and diagnostic procedures, rising healthcare expenditure, growing geriatric population, higher prevalence of lifestyle-related diseases, and rapid modernization and expansion of healthcare infrastructure.
Competitive Insight
Key players in the guidewires market are catering the demand of these devices by investing on technologically advanced products in their product portfolio across the globe. Medtronic PLC, Boston Scientific, C.R. Bard, Terumo Corporation, Abbott Laboratories, B. Braun, Johnson & Johnson, Stryker Corporation, Olympus Corporation, Angiodynamics, and Cardinal Health are the key players offering guidewires.
Terumo Medical Care Solutions is a leading manufacturer of injection and infusion therapy devices that help clinicians consistently perform at their very best.
Medtronic plc is an American-Irish medical device company. The company's operational and executive headquarters are in Minneapolis, Minnesota
Recent Developments by Key Players
Medtronic launched the Steerant Aortic Guidewire, tailored to facilitate catheter placement and exchange during diagnostic or interventional procedures in the aorta.
Terumo Cardiovascular (a global leader in cardiovascular surgery technologies) received a grant from the U.S. Food and Drug Administration (FDA) for 510(k) clearance for the CDI OneView Monitoring System. The next-generation CDI Systems platform provides visibility of key patient parameters during cardiopulmonary bypass surgery, critical to perfusion safety and improving patient outcomes.
The Guidewires Market report offers a comprehensive market segmentation analysis along with an estimation for the forecast period 2025–2030.
Segments Covered in the Report
- By Material
- Hybrid guidewire
- Nitinol guidewire
- Stainless steel guidewire
- By Type
- By Application
- Vascular
- Cardiology
- Neurology
- Urology
- Oncology
- Gastroenterology
- Otolaryngology
- By End-User
- Research laboratories & academic institutes
- Hospitals
- Diagnostic centers
- Surgical centers
- Ambulatory care centers
Geographical Segmentation
- North America
- Europe
- Germany
- U.K.
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia-Pacific (APAC)
- China
- Japan
- India
- South Korea
- Rest of Asia-Pacific
- Rest of the World (RoW)
- Brazil
- Saudi Arabia
- South Africa
- U.A.E.
- Other Countries
.png)
Source: VynZ Research
.png)
Source: VynZ Research



.png)
.png)